










 Official publication of the Czech Society of Ultrasound in Obstetrics and Gynecology.
Official publication of the Czech Society of Ultrasound in Obstetrics and Gynecology.

Objective: The etiology of Threatened Abortion is not clearly known. One of the hypotheses thought to play a role in the pathophysiology of threatened abortion is that bleeding occurring in threatened abortion causes the development of a chronic inflammatory reaction in the decidua. Because of this hypothesis, we aimed to evaluate the relationship between a threatened abortion and inflammatory markers procalcitonin, neutrophil-lymphocyte ratio (NLR) and platelet-lymphocyte ratio (PLR).
Methods: This is a prospective, controlled study. Serum procalcitonin, neutrophil-lymphocyte ratio and platelet-lymphocyte ratio of 55 pregnant women diagnosed with threatened abortion were compared with the results of 55 healthy pregnant women. ROC analysis of procalcitonin, NLR and PLR was performed for threatened abortions.
Results: Procalcitonin and NLR levels were significantly higher in the threatened abortions group than in the control group (p < 0.05). The area under the curve (AUC) in ROC analysis was within the 95% confidence interval for procalcitonin and NLR, both of which were statistically significant (p < 0.05). Procalcitonin and NLR resulted as appropriate parameters as diagnostic decision-makers in predicting threatened abortions.
Conclusion: There is an association between procalcitonin and NLR and threatened abortion. This result may support that inflammatory reaction plays a role in the etiology of threatened abortion.
Threatened abortions are indicated by vaginal bleeding that occurs before the 20th gestational week without cervical dilatation and without disruption of fetal or embryonic integrity (1). Vaginal bleeding occurs in 25% of all pregnancies and pregnancy loss occurs in approximately 50% of these (2).
The etiology of threatened abortion is not known exactly. There are hypotheses that suggest that the bleeding that occurs in threatened abortion triggers a variety of mechanisms. One of these hypotheses is that uterotonic mediators, which occur with bleeding, disrupt the contractile-relaxing balance in the uterus (3). Another hypothesis is that this bleeding causes the development of a chronic inflammatory reaction in the decidua. In fact, it is thought that this chronic inflammation suggested in this hypothesis causes placental developmental disorder and causes obstetric complications (premature-preterm rupture of membranes, preterm labor, preeclampsia) in the later stages of pregnancy (4).
Procalcitonin consists of 116 amino acids and is the precursor of calcitonin. It is present at immeasurably low levels in healthy people (5). Procalcitonin, which is defined as a marker of bacterial infection, draws attention with significant elevation, albeit at low levels, in some diseases (end-stage renal disease, congestive heart failure, cirrhosis, acute kidney injuries, intracerebral hemorrhages, post-operative anastomotic leaks) in which inflammatory reactions play a role, as shown in recent studies (6-8).
The precursor cells of the immune system are neutrophils. However, studies have shown that platelets also have a critical role in the immune response (9). Like neutrophils, platelets increase cytokine production at the onset of inflammation. These cytokines also stimulate new neutrophil and platelet synthesis, leading to increased inflammation (10). In many clinical studies, it has been shown that Neutrophil Lymphocyte Ratio (NLR) and Platelet Lymphocyte Ratio (PLR) are associated with many diseases in which there is no infection but where inflammation plays a role in the pathophysiology (11).
In this study, we wanted to evaluate the relationship between threatened abortion, which is thought to play a role in the pathophysiology of chronic inflammation, and the inflammatory markers procalcitonin, NLR and PLR. Since there are not many studies on this subject, we think that our study will contribute to the literature.
Ethics committee approval for the study was obtained from the ethics committee of Amasya University (protocol #34). The study and control groups were selected from pregnant women who applied to the Amasya Sabunçuoğlu Şerefeddin Training and Research Hospital Gynecology and Obstetrics clinic between 15 June 2020 and 30 November 2020. The study was designed as a prospective case-control study. In this period, 55 patients diagnosed with threatened abortion formed the study group, while 55 healthy pregnancies formed the control group. The pregnant women in the control group have consisted of those who completed their 20th week without any complaints in the follow-up. These two groups were compared in terms of demographic characteristics (age, height, weight, body mass index, educational status, parity, previous surgery), serum procalcitonin, neutrophils, lymphocytes, platelets, NLR and PLR.
Pregnant women with vaginal bleeding, no cervical dilation, and fetal heartbeat on ultrasonography between 6-20 weeks were included in the threatened abortion group. The first healthy 55 pregnant women who came to the outpatient clinic for normal control, between the 6th and 20th weeks, formed the control group. Those with hypertension, diabetes, thrombophilia, those diagnosed with hyperemesis gravidarum, those with a history of threatened abortion, preeclampsia, gestational diabetes, hellp syndrome in previous pregnancy, those with autoimmune, renal, cardiac or hepatic disease, those with diseases that may trigger inflammation (such as infectious diseases, collagen tissue disease) and those with fetal anomalies were not included in the study and control groups. Even if bleeding of patients in the study group recurred, each patient was included in the study once. The patients in the threatened abortion group who had spontaneous abortion were excluded from the study group. Patients who met the study criteria were included in the study after they were informed about the study and their consent was obtained.
Obstetric evaluation of patients was performed by a single clinician (S.M.A.) using a Mindray DC-7 ultrasound device. Week of gestation, amniotic fluid measurement, heartbeat and the presence of any hemorrhage area were evaluated for all patients.
Age, last menstrual period, parity, height, weight, education level and previous operations of all pregnant women were noted. Blood was drawn into a hemogram tube for serum neutrophil, lymphocyte and platelet levels, NLR and PLR, and into a biochemistry tube to evaluate parameters such as serum procalcitonin level, blood glucose, and renal and liver functions.
Neutrophil, lymphocyte and thrombocyte levels were evaluated with laser optics (X N-1000, Sysmex, Japan), and procalcitonin with electrochemiluminescence immunoassay (Cobas e 411, Roche, Japan).
Serum neutrophil, lymphocyte, platelet, NLR, PLR and procalcitonin levels of both groups were compared. ROC analysis was performed in terms of NLR, PLR and procalcitonin for threatened abortions.
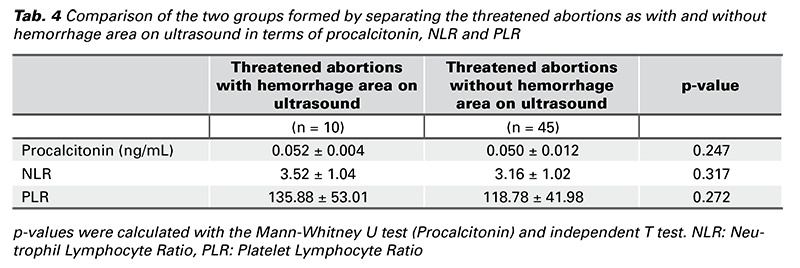
The threatened abortion group was divided in two according to the presence of hemorrhage area in ultrasonography and these two groups were also compared in terms of NLR, PLR and procalcitonin.
Power Analysis of the Study: G*Power 3.1 program was used to determine the sample of the study. The sample of our study was calculated considering that Paccolat et al. (12) evaluated procalcitonin levels during pregnancy, childbirth and postpartum period. Accordingly, the effect width was determined as d = 0.7 and according to the double-tailed hypothesis method, the confidence interval was 95% and the margin of error was 5%. As a result of the calculation, it was determined that there should be a total of 110 women, 55 for the control group and 55 for the experimental group.
Statistical analysis: Data were analyzed with IBM SPSS V23. Compliance with normal distribution was evaluated by Levene’s test. Chi-square test and Fisher’s Exact test were used to compare categorical variables in the groups. Mann-Whitney U test was used to compare non-normally distributed data in paired groups and independent two-sample t-test was used to compare normally distributed data. ROC analysis was used to determine the cut-off values for procalcitonin, NLR and PLR for threatened abortions. P < 0.05 was considered statistically significant.
All pregnant women included in the study had a live birth (premature/mature/postmature birth) except those who aborted.
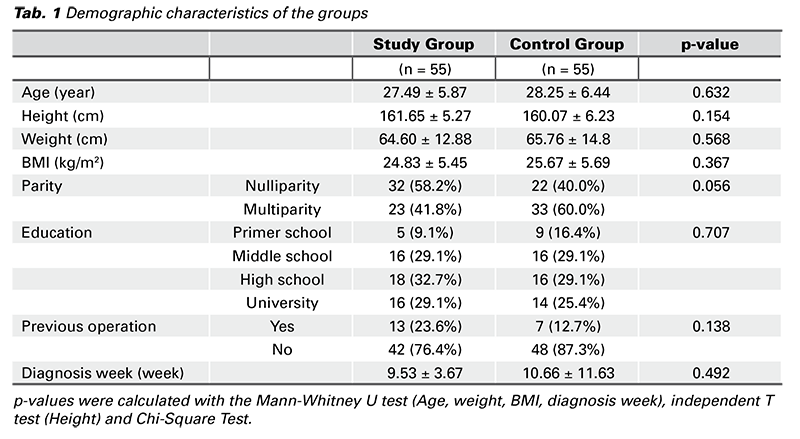
The groups were homogeneous in terms of demographic characteristics (age, parity, diagnosis week, height, weight, body mass index (BMI), educational status, previous surgery) (p > 0.05) (Tab. 1).
The comparison of the groups in terms of serum procalcitonin, neutrophils, lymphocytes, platelets, NLR and PLR is given in Tab. 2. Serum procalcitonin level was 0.051 ± 0.01 ng/mL in the threatened abortion group and 0.043 ± 0.01 ng/mL in the healthy pregnancy group. Procalcitonin level was significantly higher in the threatened abortion group than in the control group (p = 0.037).
While the mean neutrophil count was 6.46 ± 1.83×109/L in the threatened abortion group, it was 5.98 ± 1.93×109/L in the control group. The mean lymphocyte count was 2.26 ± 1.81×109/L in the threatened abortion group and 2.16 ± 0.70×109/L in the control group. The mean platelet count was 236.45 ± 49.73×109/L in the threatened abortion group and 260.20 ± 63.14×109/L in the control group. There was no significant difference between the groups in terms of mean neutrophil, lymphocyte and platelet counts (p > 0.05) (Tab. 2).
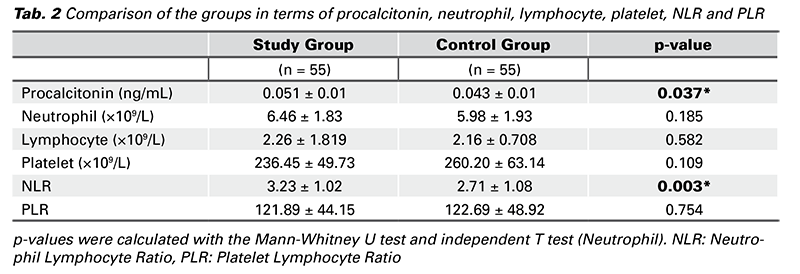
The NLR level was 3.23 ± 1.02 in the threatened abortion group and 2.71 ± 1.08 in the control group. NLR level was significantly higher in the threatened abortion group than in the control group (p = 0.003) (Tab. 2).
The PLR level was 121.89 ± 44.15 in the threatened abortion group and 122.69 ± 48.92 in the control group. There was no significant difference between the groups in terms of PLR (p = 0.754) (Tab. 2).
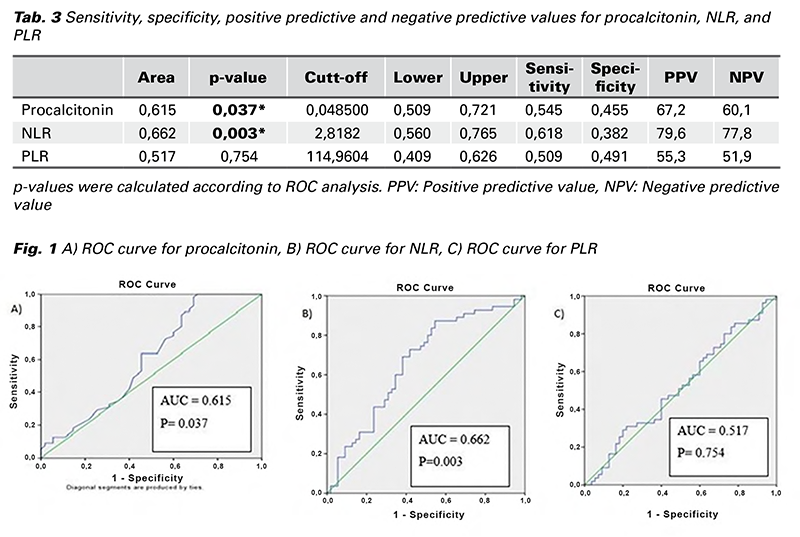
The sensitivity, specificity, positive predictive and negative predictive values of the patients for procalcitonin, NLR and PLR are given in Tab. 3. As can be seen from the table, the area under the curve (AUC) for the ROC curve (Receiver Operator Characteristics Curve) was statistically significant at the 95% confidence interval for procalcitonin and NLR (p < 0.05). These parameters had diagnostic value in predicting threatened abortions. However, the area under the ROC curve for PLR was not within the confidence interval and it did not have statistical significance (p > 0.05). This parameter had no diagnostic value in predicting threatened abortions (Tab. 3) (Fig. 1).
The threatened abortion group was divided in two according to the presence of hemorrhage area in ultrasonography and these were compared in terms of serum procalcitonin levels, NLR and PLR. Serum procalcitonin levels, NLR and PLR values were higher in pregnant women with a diagnosis of threatened abortion with hemorrhage area on ultrasonography than those without bleeding area, but there was no significant difference between the groups (p > 0.05) (Tab. 4).
One of the hypotheses of factors thought to play a role in the pathophysiology of threatened abortion is the chronic inflammatory reaction that develops in the decidua with bleeding. Determining the relationship between threatened abortion and inflammatory markers may support the validity of this proposed hypothesis. Thus, it may be beneficial to clarify the etiology of threatened abortion and to clarify the complications this may cause in the later stages of pregnancy. Therefore, in this study, we evaluated the relationship between threatened abortion and the inflammatory markers procalcitonin, NLR and PLR.
Threatened abortions are a fairly common complication of pregnancy. They affect 25% of pregnancies (13). In addition to affecting the physical health of pregnant women, they also have negative effects on their psychological state as they create emotional stress. Although it is seen so frequently, the causative factors and mechanism have not been fully explained. Therefore, threatened abortions are difficult to predict and predictive methods are unreliable (14). It can also be confused with bleeding from other causes in the early stages of pregnancy (ectopic pregnancy, incomplete abortion) (15). For this reason, the detection of new markers that contribute to illuminating the pathophysiology that causes threatened abortions and that could even help in the diagnosis of threatened abortion may provide clinical benefit. We planned our study for this purpose.
Procalcitonin level is normally < 0.5 ng/mL (16). The rise of procalcitonin occurs immediately due to its cytokine- like behavior (17). It reaches detectable levels in 4 hours and peaks at 6 hours (18). Although the normal level is said to be < 0.5 ng/mL, the appropriate cut-off values for procalcitonin differ according to the reason in recent studies. For example, serum levels in patients with heart failure were 0.06 ng/mL (19), while serum levels above 0.05 in patients with cirrhosis were found to be significant (8).
There are studies evaluating normal serum procalcitonin levels during pregnancy. Paccolat et al. reported that serum procalcitonin level was 0.043 μg/L at 24-28 weeks of gestation. They stated that this level increased in the later weeks of pregnancy, and was 0.068 μg/L at birth, 0.200 μg/L on postpartum day 2-3, and 0.060 μg/L on postpartum day 10 (12). Similar to this study, we found the serum procalcitonin level to be 0.043 ± 0.01 ng/mL in healthy pregnant women in our study. In the threatened abortions group, it was 0.051 ± 0.01 ng/mL and this was significantly higher than the healthy pregnancy group. Procalcitonin was significant as a diagnostic decision maker in predicting threatened abortion.
Studies have shown that various obstetric and neonatal complications develop in the following weeks in pregnant women with threatened abortion (20). Clarifying its pathophysiology would be beneficial in taking precautions against these complications and reducing maternal/fetal mortality/morbidity. Therefore, it is important to determine the causes that play a role in its etiology.
In the literature, the evaluation of NLR and PLR in threatened abortion has been compared with pregnancies resulting in abortion. Thus, their use as precursors in early pregnancy was investigated (21,22). Studies are rare comparing threatened abortions and healthy pregnancies in terms of NLR and PLR. One of them is the work of Ata et al. From the results of this study, no significant difference was found between the groups in terms of NLR, but they reported that the level of PLR in the threatened abortions group was higher than the healthy pregnancy group (23). In our study, contrary to the results of this study, the NLR level in the threatened abortion group was significantly higher than the healthy pregnancy group, but there was no significant difference between the groups in terms of PLR. In ROC analysis, NLR was statistically significant at the 95% confidence interval, showing it to be useful as a diagnostic decision maker in predicting threatened abortion. However, there was no significance in ROC analysis for PLR.
There have been studies evaluating other inflammatory markers that determined that an inflammatory process plays a role in the etiopathogenesis of threatened abortion. In the study by Keskin et al., vascular endothelial growth factor (VEGF), soluble VEGF receptor-1 (sVEGFR-1) and VEGF/sVEGFR-1 were found to be associated with the pathogenesis of threatened abortion (24). The relationship of many immune and inflammatory markers (apolipoprotein A-1, IL2β receptor, IL6, IL8, IL10, IL1β and TNFα) with threatened abortion has been determined (25,26). In a review that was conducted, it is stated that serum Ca 125 is a valuable predictive marker for determining ‚probable to continue‘ pregnancies (27). As in our study, these studies also confirm the presence of inflammation in the etiopathogenesis of threatened abortion.
The presence of a biomarker that determines whether a pregnancy will continue or will result in abortion may be beneficial in terms of providing preliminary information to the expectant mother with increased anxiety and whether the drugs used in the treatment of threatened abortion should be continued. Although it does not affect the treatment process for threatened abortion, it may be important in terms of exemption from the side effects and cost of the drugs used in the treatment.
In our study, we also compared the cases with a hemorrhage area on ultrasonography in the threatened abortion group with those without a hemorrhage area. As a result of this comparison, procalcitonin, NLR and PLR were higher in the group with hemorrhage area, but this was not significant. Although not statistically significant, the fact that these parameters were higher in the group compared to those without hemorrhage area may be due to the persistence of this hemorrhage detected by ultrasound for a certain period of time, triggering the chronic inflammatory reaction in the decidua with bleeding, which is one of the hypotheses put forward in the pathophysiology of threatened abortion (4).
The limitation of our study was that the number of cases was as high as the number detected in the power analysis. Nevertheless, if we had more cases, it would have increased the power of our study. In the study, the threatened abortion group was compared only with the healthy pregnancy group. Since the number of pregnant women who had spontaneous abortions after threatened abortions was low, we could not include them in the study. Our study would be more meaningful if we could compare this abortion group with the healthy pregnancy group with threatened abortions. The strength of our study was that serum procalcitonin, NLR and PLR were studied simultaneously in threatened abortions, and this is rare.
In conclusion, procalcitonin and NLR levels in threatened abortions were higher than in healthy pregnancies. According to this result, it can be said that there is a relationship between threatened abortion and the inflammatory markers procalcitonin and NLR. In addition, the increase in procalcitonin and NLR during pregnancy may indicate an increased risk of threatened abortion.